Substrate-dependent regiodivergence in [3 + 2] annulation reactions of 2-(phenacylethylidene)cyclobutanones with thioureas
IF 2.9
3区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
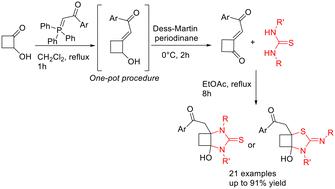
The [3 + 2] annulation reaction between a thiourea, an ambident dinucleophile, and a 2-(phenacylethylidene)cyclobutanone, containing a novel pull–pull alkene system, could in principle proceed with several chemo- and regioselectivity profiles. Here we describe a convenient synthesis of the functionalized cyclobutanone substrates and show that they react with thioureas in a manner that is rationalized mechanistically in terms of the steric and electronic effects at play. The [3 + 2] annulation proceeds in mild, additive-free conditions to provide access to previously unknown cyclobutane-fused imidazolidine-2-thione and thiazolidine-2-imine derivatives in good yields.
2-(苯基亚乙基)环丁酮与硫脲的[3 + 2]环化反应中的底物依赖性变构
原则上,硫脲、含歧二核亲和剂和含有新型拉拉烯系统的 2-(苯乙亚基)环丁酮之间的 [3 + 2] 环化反应可以通过几种化学和区域选择性曲线进行。在这里,我们描述了官能化环丁酮底物的简便合成方法,并表明它们与硫脲类化合物的反应方式从机理上讲是合理的,即立体效应和电子效应在起作用。[3+2]环化反应在温和、无添加剂的条件下进行,从而以良好的产率获得了以前未知的环丁烷融合咪唑烷-2-硫酮和噻唑烷-2-亚胺衍生物。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Organic & Biomolecular Chemistry
化学-有机化学
CiteScore
5.50
自引率
9.40%
发文量
1056
审稿时长
1.3 months
期刊介绍:
The international home of synthetic, physical and biomolecular organic chemistry.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


