Telluride-Based Pillar[5]arene: A Recyclable Catalyst for Alkylation Reactions in Aqueous Solution.
IF 3.3
2区 化学
Q1 CHEMISTRY, ORGANIC
The Journal of Organic Chemistry
Pub Date : 2024-09-20
Epub Date: 2024-09-04
DOI:10.1021/acs.joc.4c00997
引用次数: 0
Abstract
The syntheses of previously unknown sulfide- and telluride-pillar[n]arenes are reported here. These macrocycles, among others, were tested as catalysts for alkylation reactions in aqueous solutions. Telluride-pillar[5]arene (P[5]-TePh) showed the best performance, emulating the behavior of the methyltransferase enzyme cofactor S-adenosyl-l-methionine. Using 1.0 mol % of P[5]-TePh, benzyl bromides reacted with NaCN/NaN3 in water, yielding organic nitriles/azides. The catalyst was recycled and efficiently reused for up to six cycles. 1H NMR experiments indicate a possible interaction between the substrate and P[5]-TePh's cavity.
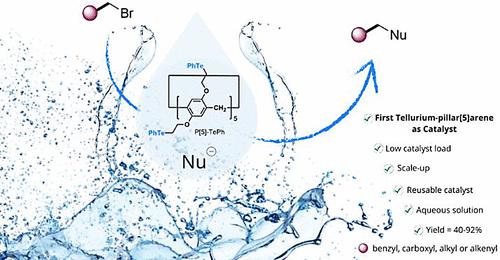
碲基支柱[5]炔:水溶液中烷基化反应的可循环催化剂。
本文报道了之前未知的硫化物和碲化物柱[n]烯的合成。除其他外,还测试了这些大环作为催化剂在水溶液中进行烷基化反应的情况。碲-支柱[5]炔(P[5]-TePh)表现出了最佳性能,模仿了甲基转移酶辅助因子 S-腺苷-l-蛋氨酸的行为。使用 1.0 mol % 的 P[5]-TePh,苄基溴与 NaCN/NaN3 在水中发生反应,生成有机腈/氮化物。催化剂可回收并有效重复使用长达六个循环。1H NMR 实验表明,底物与 P[5]-TePh 的空腔之间可能存在相互作用。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

The Journal of Organic Chemistry
化学-有机化学
CiteScore
6.20
自引率
11.10%
发文量
1467
审稿时长
2 months
期刊介绍:
The Journal of Organic Chemistry welcomes original contributions of fundamental research in all branches of the theory and practice of organic chemistry. In selecting manuscripts for publication, the editors place emphasis on the quality and novelty of the work, as well as the breadth of interest to the organic chemistry community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


