Combinatorial quantification of 5mC and 5hmC at individual CpG dyads and the transcriptome in single cells reveals modulators of DNA methylation maintenance fidelity
IF 12.5
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
Inheritance of 5-methylcytosine from one cell generation to the next by DNA methyltransferase 1 (DNMT1) plays a key role in regulating cellular identity. While recent work has shown that the activity of DNMT1 is imprecise, it remains unclear how the fidelity of DNMT1 is tuned in different genomic and cell state contexts. Here we describe Dyad-seq, a method to quantify the genome-wide methylation status of cytosines at the resolution of individual CpG dinucleotides to find that the fidelity of DNMT1-mediated maintenance methylation is related to the local density of DNA methylation and the landscape of histone modifications. To gain deeper insights into methylation/demethylation turnover dynamics, we first extended Dyad-seq to quantify all combinations of 5-methylcytosine and 5-hydroxymethylcytosine at individual CpG dyads. Next, to understand how cell state transitions impact maintenance methylation, we scaled the method down to jointly profile genome-wide methylation levels, maintenance methylation fidelity and the transcriptome from single cells (scDyad&T-seq). Using scDyad&T-seq, we demonstrate that, while distinct cell states can substantially impact the activity of the maintenance methylation machinery, locally there exists an intrinsic relationship between DNA methylation density, histone modifications and DNMT1-mediated maintenance methylation fidelity that is independent of cell state. Here the authors develop a method to quantify all combinations of 5-methylcytosine (5mC) and 5-hydroxymethylcytosine at individual CpG dyads, including in single cells, to identify the relationship between the local 5mC density, histone marks and maintenance methylation fidelity.
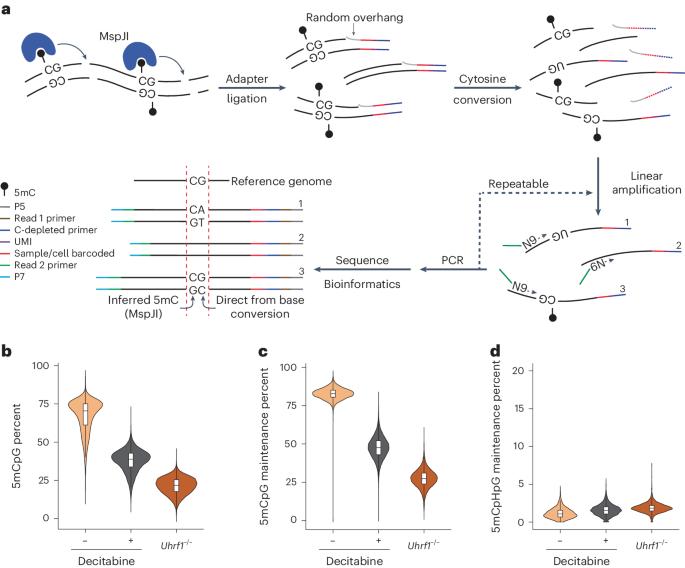
单个细胞中单个 CpG 二元组和转录组上 5mC 和 5hmC 的组合量化揭示了 DNA 甲基化维持保真度的调节因子
DNA 甲基转移酶 1(DNMT1)将 5-甲基胞嘧啶从一代细胞遗传到下一代细胞,在调节细胞特性方面起着关键作用。虽然最近的研究表明 DNMT1 的活性并不精确,但仍不清楚 DNMT1 的保真度在不同的基因组和细胞状态下是如何调整的。在这里,我们描述了 Dyad-seq,这是一种以单个 CpG 二核苷酸为分辨率量化全基因组胞嘧啶甲基化状态的方法,发现 DNMT1 介导的维持甲基化的保真度与 DNA 甲基化的局部密度和组蛋白修饰景观有关。为了更深入地了解甲基化/去甲基化的周转动态,我们首先扩展了 Dyad-seq 来量化单个 CpG 二聚体上 5-甲基胞嘧啶和 5-羟甲基胞嘧啶的所有组合。接下来,为了了解细胞状态转变如何影响甲基化的维持,我们缩小了该方法的规模,以联合剖析全基因组甲基化水平、甲基化维持保真度和来自单细胞的转录组(scDyad&T-seq)。利用 scDyad&T-seq,我们证明,虽然不同的细胞状态会对甲基化维持机制的活性产生重大影响,但在 DNA 甲基化密度、组蛋白修饰和 DNMT1 介导的甲基化维持保真度之间存在着与细胞状态无关的内在联系。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature Structural & Molecular Biology
BIOCHEMISTRY & MOLECULAR BIOLOGY-BIOPHYSICS
CiteScore
22.00
自引率
1.80%
发文量
160
审稿时长
3-8 weeks
期刊介绍:
Nature Structural & Molecular Biology is a comprehensive platform that combines structural and molecular research. Our journal focuses on exploring the functional and mechanistic aspects of biological processes, emphasizing how molecular components collaborate to achieve a particular function. While structural data can shed light on these insights, our publication does not require them as a prerequisite.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


